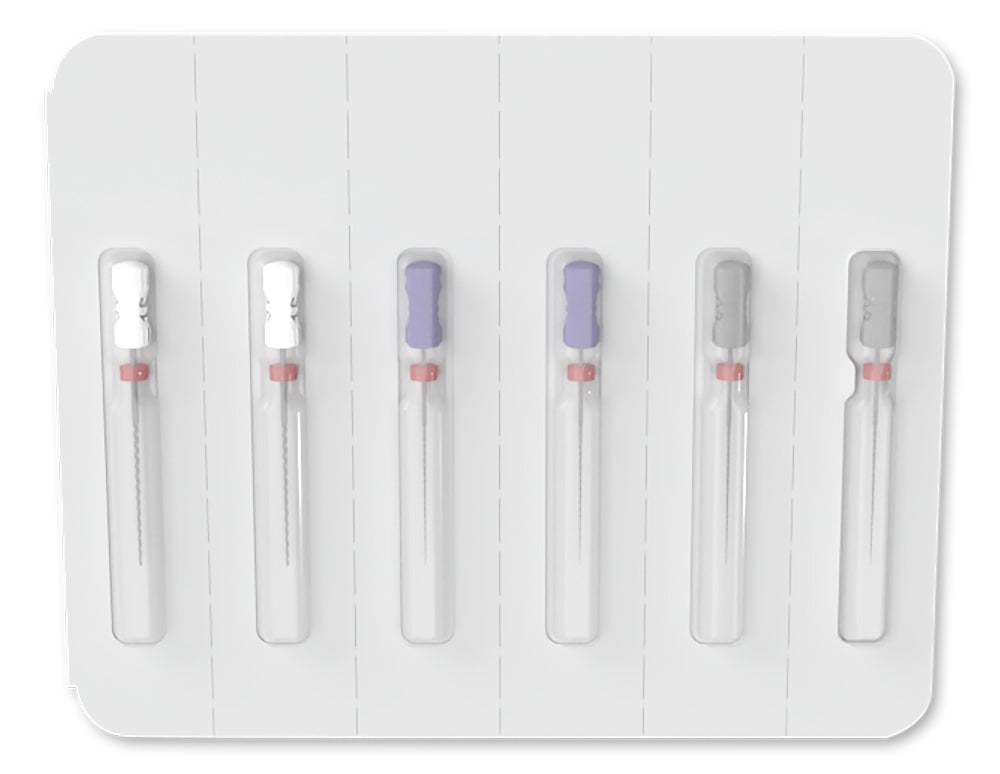
MicroMega K-Files Sterile Hand Files
MicroMega K-Files Sterile Hand Files
SKU:910-60029717
Couldn't load pickup availability
Tactile anatomy scouting for confident canal preparation.
MicroMega K-Files are stainless steel endodontic hand files used to scout the root canal before shaping. Worked into the canal, they give a first tactile read on anatomy, mineralization, and curvature — the information clinicians need to gauge case difficulty and choose the right NiTi sequence before shaping begins.
Use them as the primary instrument in fully manual root canal treatment, or as a supplement in mechanized workflows: scout first with the K-File, then shape with NiTi. The 0.02 taper and ISO-standard tip sizes pair predictably with virtually any rotary or reciprocating system.
MicroMega is the endodontic instrument brand of Coltène/Whaledent, manufactured by Micro-Mega SA in Besançon, France.
Features
- Stainless steel construction with 0.02 (2%) taper
- ISO color-coded handles for at-a-glance size identification
- Pre-mounted radiopaque silicone stops
- Handle marking to indicate pre-curvature orientation
- Pre-sterilized in blisters of 6 — ready to open and use chairside
- Multi-use: reprocess and re-sterilize per manufacturer's IFU
Clinical Uses
- Scouting and evaluating canal anatomy, curvature, and mineralization
- Determining working length, with or without an electronic apex locator
- Removing initial mechanical constraints through controlled enlargement
- Supplementing NiTi files in mechanized workflows
Specifications
- Material: Stainless steel
- Taper: 0.02 (2%)
- ISO sizes: 006 through 080, plus assorted packs (008/015, 015/040, 045/080)
- Lengths: 21 mm, 25 mm, 31 mm
- Pack quantity: Blister of 6 sterile instruments
- Sterilization: Pre-sterilized; multi-use
- Manufacturer: Micro-Mega SA (Coltène Group), France
Medical device for professional dental use only. CE-marked Class Is per Directive 93/42/EEC. Refer to the manufacturer's instructions for use for full reprocessing and clinical guidance.